Researchers are still attempting to identify a correlation between mercury poisoning and youth conditions such as autism, ADHD, and SID, all of which affect a child’s ability to concentrate. Continued studies have been conducted on the effects of thimerosal, many addressing its possible neurological effects. The study did not uncover any evidence that ethylmercury caused health risks to children, but in an overall effort to limit mercury use, medicine suppliers were asked to reduce or discontinue use of the product.

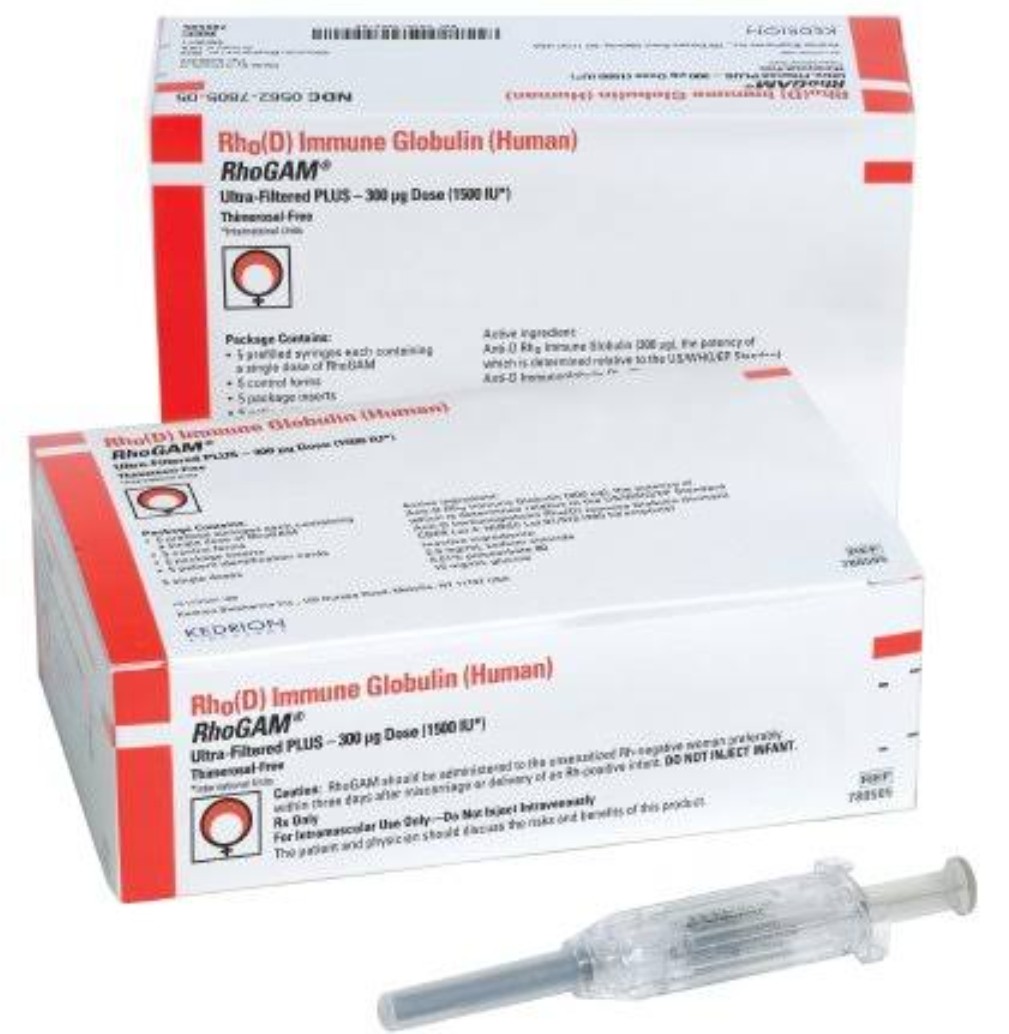
In 1999, the FDA conducted a study on the effects of thimerosal on children. Ethylmercury has not been shown to be as toxic as methylmercury but, in large doses, can have effects ranging from local allergic reactions to central nervous system damage. However, the preservative’s ethylmercury content has been scrutinized because numerous studies have shown that methylmercury, a separate organomercurial composition, causes neurological disorders, especially in young children and infants in utero. Thimerosal has been shown to be effective in combating pathogens. Thimerosal Preservative and RhoGAM®īetween its introduction onto the market and April of 2001, RhoGAM® contained the preservative thimerosal. The foreign fetal cells are coated in the RhoGAM® antibodies and expulsed from the woman’s system before her body can detect them. The antibodies in the RhoGAM® attack any fetal blood cells that are outside the placenta. The treatment is a brand of Rh immunoglobulin (an immunoglobulin is a protein released by cells that functions in immune responses by bonding to antigens or foreign matter). The RhoGAM® treatment is derived from human plasma and contains antibodies that Rh-negative blood donors have created in response to encounters with Rh-positive blood. Rh-negative women are given a RhoGAM® injection during the twenty-eighth week of pregnancy and again after delivery. The treatment keeps an Rh-negative woman from creating antibodies to the Rh-positive blood of her child. RhoGAM® was first made available in 1968.
